The Michael Barber Centre supports the research activities of a number of groups.
Perdita Barran’s Research Group https://www.mbc.manchester.ac.uk/barrangroup/
 We develop mass spectrometry based methods and instruments to allow us to look at conformation, conformational change and aggregation. Methods that allow us to preserve non-colvalent protein complexes are also applied to inorganic supramolecular biomimetic systems. We also generate IM-MS data from standard small molecules and proteins that can be used to calibrate TW IM-MS instrumentation. Our principal areas of research interest are in understanding pre-fibrillar aggregation, intrinsically disordered proteins and how to tame disorder, probing the stability of protein complexes and of model peptide systems.
We develop mass spectrometry based methods and instruments to allow us to look at conformation, conformational change and aggregation. Methods that allow us to preserve non-colvalent protein complexes are also applied to inorganic supramolecular biomimetic systems. We also generate IM-MS data from standard small molecules and proteins that can be used to calibrate TW IM-MS instrumentation. Our principal areas of research interest are in understanding pre-fibrillar aggregation, intrinsically disordered proteins and how to tame disorder, probing the stability of protein complexes and of model peptide systems.
Molecular Magnetism group – David Collison, Eric McInnes & Richard Winpenny http://www.molmag.manchester.ac.uk/
 The group are world-leaders in the synthesis and characterisation of molecular magnets, ranging from studies of complexes of highly anisotropic metal ions for information storage, to use of heterometallic rings as qubits for quantum information processing. The heterometallic rings are superb subjects for mass spectrometry, with individual rings showing sharp well-resolved MS by electrospray methods. More recently we have been working with the Michael Barber centre to study very high mass (15 kD) assemblies of rings, showing that these supramolecular compounds that rival small proteins in size can be studied by advanced mass spectrometry.
The group are world-leaders in the synthesis and characterisation of molecular magnets, ranging from studies of complexes of highly anisotropic metal ions for information storage, to use of heterometallic rings as qubits for quantum information processing. The heterometallic rings are superb subjects for mass spectrometry, with individual rings showing sharp well-resolved MS by electrospray methods. More recently we have been working with the Michael Barber centre to study very high mass (15 kD) assemblies of rings, showing that these supramolecular compounds that rival small proteins in size can be studied by advanced mass spectrometry.
Surface Analysis Research Centre – Nick Lockyer http://www.sarc.manchester.ac.uk
 The group research interests focus on the development and application of surface analysis and chemical imaging, particularly Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS) and laser post-ionisation. There are three themes to this research: fundamentals, instrument development and novel applications. The Life Sciences provide an exciting field in which to develop new applications, which in turn drive developments in methodology and technology. Ongoing activities include the study of cluster primary ion impacts on molecular surfaces, the development of new ToF-SIMS instrumentation, and methodology research into ToF-SIMS imaging of biological cells and tissue. We are currently seeking to exploit these advances through a variety of collaborations with fellow academics, industrial partners and healthcare professionals.
The group research interests focus on the development and application of surface analysis and chemical imaging, particularly Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS) and laser post-ionisation. There are three themes to this research: fundamentals, instrument development and novel applications. The Life Sciences provide an exciting field in which to develop new applications, which in turn drive developments in methodology and technology. Ongoing activities include the study of cluster primary ion impacts on molecular surfaces, the development of new ToF-SIMS instrumentation, and methodology research into ToF-SIMS imaging of biological cells and tissue. We are currently seeking to exploit these advances through a variety of collaborations with fellow academics, industrial partners and healthcare professionals.
Sabine L Flitsch’s Research Group http://www.flitschlab.chemistry.manchester.ac.uk/
 Our research interests are on the interface of Chemistry and Biology with focus on applications in Biotechnology. A particular interest is the use of biocatalysis for the synthesis of complex carbohydrates and glycoconjugates such as glycolipids, glycoproteins, polysaccharides, glycomaterials and glycoarrays. These glycoarrays are used to discover new carbohydrate-protein interactions using mass spectrometry (MALDI-Tof) as a label-free analytical tool. More recently, we have started using ion mobility mass spectrometry (in collaboration with Profs Eyers and Barran) for sequencing of complex carbohydrates.
Our research interests are on the interface of Chemistry and Biology with focus on applications in Biotechnology. A particular interest is the use of biocatalysis for the synthesis of complex carbohydrates and glycoconjugates such as glycolipids, glycoproteins, polysaccharides, glycomaterials and glycoarrays. These glycoarrays are used to discover new carbohydrate-protein interactions using mass spectrometry (MALDI-Tof) as a label-free analytical tool. More recently, we have started using ion mobility mass spectrometry (in collaboration with Profs Eyers and Barran) for sequencing of complex carbohydrates.
Amin Rostami’s Research Group http://www.mcisb.org/syspharm/
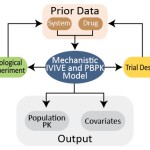 The systems pharmacology group seeks to establish proper quantitative in vitro to in vivo extrapolation (IVIVE) to predict in vivo consequences upon administration of drugs to humans. Such IVIVE requires measurement of the abundance of relevant enzymes, transporters and receptors. These data will enable physiologically based computer models of molecular networks. In the first instance, this group has established robust quantitative methods to measure abundance of human drug metabolising enzymes and transporters in relevant human and animal tissues and in vitro cell lines. The quantification of the proteins that play an essential role in the metabolism and disposition of drugs and other xenobiotics in different organs is now underway and the group has several running projects for various tissues including the liver, the intestine, the kidneys, and the brain. Other areas of interest to the group include quantification of the expression of enzymes and transporters in paediatric subjects and in disease states, particularly cancer. The methods employed are based on the QconCAT technology using selected reaction monitoring (SRM) assays on nanoLC-ESI-QqQ.
The systems pharmacology group seeks to establish proper quantitative in vitro to in vivo extrapolation (IVIVE) to predict in vivo consequences upon administration of drugs to humans. Such IVIVE requires measurement of the abundance of relevant enzymes, transporters and receptors. These data will enable physiologically based computer models of molecular networks. In the first instance, this group has established robust quantitative methods to measure abundance of human drug metabolising enzymes and transporters in relevant human and animal tissues and in vitro cell lines. The quantification of the proteins that play an essential role in the metabolism and disposition of drugs and other xenobiotics in different organs is now underway and the group has several running projects for various tissues including the liver, the intestine, the kidneys, and the brain. Other areas of interest to the group include quantification of the expression of enzymes and transporters in paediatric subjects and in disease states, particularly cancer. The methods employed are based on the QconCAT technology using selected reaction monitoring (SRM) assays on nanoLC-ESI-QqQ.

